Synthesis and performance of graphene and activated carbon composite for absorption of three-valance arsenic from wastewater
Vol 2, Issue 01, Pages 63-72,*** Field:Nanotechnology in Environmental Health
Abstract
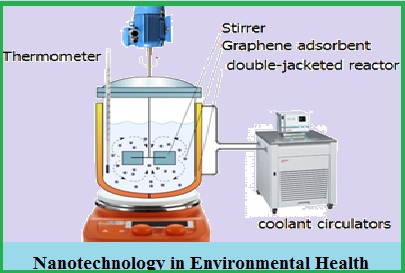
High levels of arsenic in the effluent are a major concern of human, and the removal of it from the wastewater is hard and costly. The most common techniques for removal of arsenic are membrane separation, ion exchange, oxidation, and coagulation. The active carbon is used as the most common arsenic adsorbent in wastewater treatment processes, this study has been considered as the main adsorbent and attempted to improve its surface properties by graphene nanosheets. Thus, by adding graphene (4.5%w) to the carbon structure, its porosity increases, and the ion exchange behavior and surface load are corrected. In this research, the effects of time process, concentration of arsenic, and adsorbent are evaluated in different pH values. It has been observed that the maximum adsorption of arsenic is 91.8%; in addition, when graphene is used, the rate of absorption of Arsenic has increased about 5.2%, and the process time is shortened.
References
M. A. P. Cechinel, A. A. U. de Souza, Study of arsenic adsorption onto activated carbon originating from cow bone, J. Clean. Prod., 65 (2014) 342–349.
J. H. Park, Y. S. Han, J. S. Ahn, Comparison of arsenic co-precipitation and adsorption by iron minerals and the mechanism of arsenic natural attenuation in a mine stream, Water Res., 106 (2016), 295–303.
L. R. Molinari, P. Argurio, Arsenic removal from water by coupling photocatalysis and complexation-ultrafiltration processes: A preliminary study, Water Res., 109 (2017) 327-336.
B. Pakzadeh, J. R. Batista, Surface complexation modeling of the removal of arsenic from ion-exchange waste brines with ferric chloride, J. Hazard. Mater., 188 (2011) 399–407.
D. Santra, M. Sarkar, Optimization of process variables and mechanism of arsenic (V) adsorption onto cellulose nanocomposite, J. Mol. Liquids, 224 (2016) 290–302.
O. P. Chen, Y. J. Lin, W. Z. Cao, C. T. Chang, Arsenic removal with phosphorene and adsorption in solution, Mater. Lett., 190 (2017) 280–282.
A. Sigdel, J. Park, H. Kwak, P. Pyung-Kyu, Arsenic removal from aqueous solutions by adsorption onto hydrous iron oxide-impregnated alginate beads, J. Ind. Eng. Chem., 35 (2016) 277–286.
A.A.H. Izzeldin, S.M. Bice, J. Catherine, O.N. Vincent, Adsorption studies of aqueous Pb(II) onto a sugarcane bagasse/multi-walled carbon nanotube composite, Phys. Chem. Earth 66 (2013) 157–166.
R. Soni Dericks, P. Shukla, Data on Arsenic(III) removal using zeolite-reduced graphene oxide composite, Data in Brief, 22 (2019) 871-877.
G. Zhao, X. Ren, X. Gao, X. Tan, J. Li, C. Chen, Y. Huang, X. Wang, Removal of Pb(II) ions from aqueous solutions on few-layered graphene oxide nanosheets, Dalton Trans., 40, (2011) 945–952.
L. Cui, Y. Wang, L. Gao, L. Hu, L. Yan, Q. Wei, B. Du, EDTA functionalized magnetic graphene oxide for removal of Pb(II), Hg(II) and Cu(II) in water treatment: adsorption mechanism and separation property, Chem. Eng. J., 281 (2015) 1–9.
Z. Goharibajestani, A. YudaYürüm, Effect of transition metal oxide nanoparticles on gas adsorption properties of graphene nanocomposites, Appl. Surface Sci., 475 (2019) 1070-1076.
G. Zhao, J. Li, X. Ren, C. Chen, X. Wang, Few-layered grapheme oxide nano-sheets as superior sorbents for heavy metal ion pollution management, Environ. Sci. Technol., 45 (2011) 454–462,
C. Zheng, X. Zhou, H. Cao, G. Wang, Z. Liu, Synthesis of porous graphene/activated carbon composite with high packing density and large specific surface area for supercapacitor electrode material, J. Power Sources, 258 (2014) 290–296.
M. Sohail, M. Saleem, S. Noor Saeed, A. Afridi, M. Khan, M. Arif, Modified and improved Hummer's synthesis of graphene oxide for capacitors applications, Modern Elec. Mater., 3 (2017) 110-116.
A. Ganesan, M. M. Shaijumon, Activated graphene-derived porous carbon with exceptional gas adsorption properties, Micropor. Mesopor. Mater., 220 (2016) 21-27.
JOURNAL PUBLISHING AGREEMENT
PLEASE PROVIDE US THE FOLLOWING INFORMATION,
Article entitled:
Corresponding author:
To be published in the journal:
Your Status
I am the sole author of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
I am one author signing on behalf of all co-authors of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
Please tick the above blanks (as appropriate), review the Journal Publishing Agreement, and then sign and date the document in black ink.
Published Journal Article: the author may share a link to the formal publication through the relevant DOI. Additionally theses and dissertations which contain embedded Published Journal Articles as part of the formal submission may be hosted publicly by the awarding institution with a link to the formal publication through the relevant DOI. Any other sharing of Published Journal Articles is by agreement with the publisher only.
Signed: ______________________________________ Name printed: ___________________________________________
Title and Company (if employer representative): _______________________Date: __________________________________